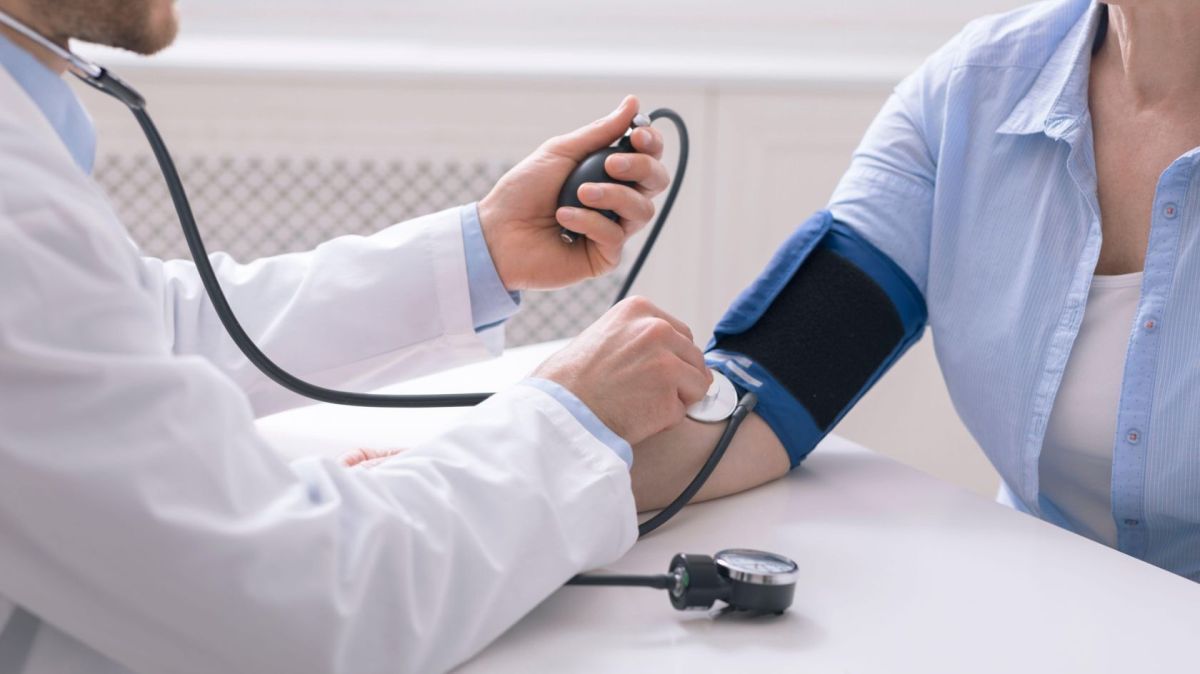
Food supplements are subject to a different regulation from medicines. While medicines are regulated by Infarmed, supplements do not need to be approved and tested by this Portuguese government agency that evaluates, authorises, regulates and controls human medicines.
And this is where the controversy begins. This lack of regulation has already made the medical association take a stand against these alternative products, arguing that there is no evidence to point to the safety of these supplements, as they are not tested before entering the market by Infarmed.
However, that doesn't prevent people from consuming them in the hopes of improving their health. According to a recent study by Deco Protest, in Portugal about four out of ten Portuguese people took supplements in the last 12 months - most take supplements to strengthen the immune system.
The conclusions of this online survey, conducted in May 2021 with samples between 18 and 74 years old, showed that after strengthening the immune system, the main reasons that leads consumers to buy these products is to increase energy levels and raise the levels of vitamins and minerals in the body.
In addition, the majority of survey respondents (71 percent) believe that supplements will boost their immune system and prevent infections like that caused by Covid-19. In fact, "three out of ten people who were already taking supplements before the pandemic increased their consumption when the SARS-CoV-2 virus appeared," said Deco Protest in a note, adding that "however, so far there is no scientific evidence to support that, which means it's no more than a myth.”
The survey was answered by 1001 Portuguese citizens. Of those, 59 percent were women, with an average age of 39 years old.
Who regulates food supplements in Portugal?
Although there is no mandatory inspection before being placed on the market from Infarmed, according to the ASAE (Economic and Food Safety Authority) the DGAV (Food and Veterinary Authority) may at any time ask the producer to send scientific papers and data that prove the compliance of the products on the market.
In addition, "economic operators must ensure that the content of food supplements complies with what is on the label and it is up to the DGAV to monitor compliance with the legal requirements of food supplements in physical and online stores".
In relation to e-commerce, this is one of the new priorities of ASAE since many of the purchases are made online. For this reason, ASAE is carrying out inspections of economic activities in a “digital environment”, with the aim of verifying compliance with the law.
According to ASAE, in an interview with The Portugal News, there are five mandatory requirements that these labels must contain: “The designation of the categories of nutrients or substances that characterise the product or specific reference to its nature; a recommended daily dose of the product; a note saying that the prescribed daily dose must not be exceeded; the indication that food supplements should not be used as a substitute for a varied diet; and a warning saying that it must be stored out of reach of children”.
Whenever products do not comply with legal requirements, they are withdrawn from the market and if they present a risk to public health, this entity will make statements to consumers regarding the risk, explained ASAE.
According to this entity, in the first half of 2021, a total of 4 320 thousand products were seized, mainly due to misrepresentation and false advertising.
One instance of misrepresentation occurs when a supplement that claims to be 100 percent natural ends up containing medicinal substances to enhance the intended effect. This is a cause of concern, first in terms of disloyalty because the customer was looking for something natural, otherwise he would have taken a medicine. Also, if the product contains drug substances, it should have been tested by Infarmed before being placed on the market.
If you find out that any food supplement does not comply with what is stated on the label, you can file a complaint using the form on the ASAE website at https://www.asae.gov.pt/denuncias1.aspx.
Paula Martins is a fully qualified journalist, who finds writing a means of self-expression. She studied Journalism and Communication at University of Coimbra and recently Law in the Algarve. Press card: 8252